1. The Diagnostic Dilemma
For most, a persistent headache is a mundane annoyance, easily muffled by ibuprofen and a dark room. But for a subset of patients, that dull throb is the first warning shot of a neurological crisis. Under the current standard of care, the path from a GP’s office to a definitive answer is a gauntlet of high-cost MRI queues and the visceral terror of a surgical biopsy—a procedure that requires drilling a hole into the skull to extract live brain tissue.
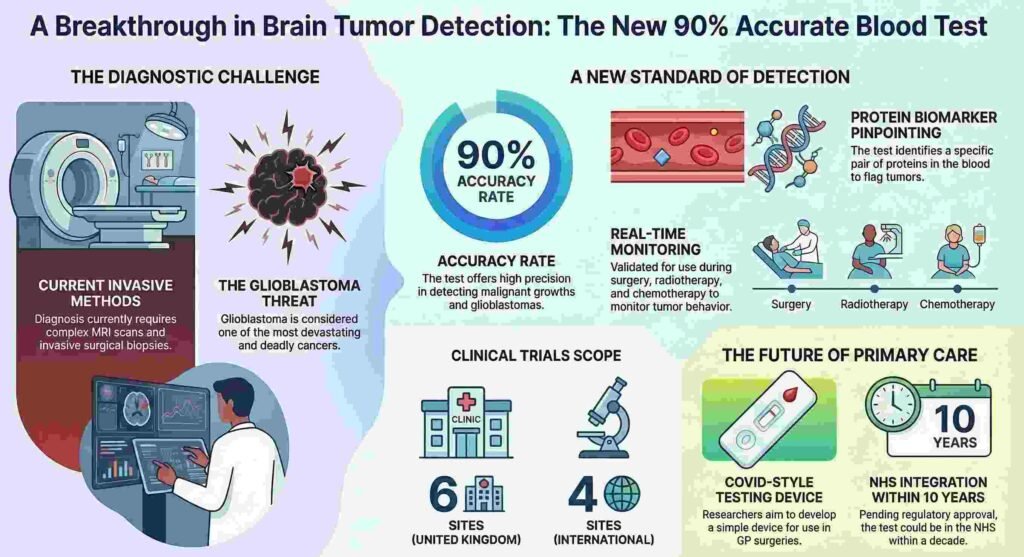
Researchers at the University of Manchester, collaborating with international teams in Denmark, have developed a technology that could dismantle this diagnostic barrier. They have successfully mapped a molecular signature in the blood that identifies malignant growths with staggering precision. This breakthrough represents a shift in oncology, moving away from reactive, invasive procedures toward a future of proactive, “liquid” diagnostics.
2. The 90% Breakthrough: Accuracy That Rivals Surgery
The Manchester-led team has decoded the biological markers of glioblastoma—the most aggressive and common primary brain cancer—with a test that is over 90% accurate. This is not merely an incremental improvement; it is a disruptive leap in how we identify “deadly” malignancies.
The science hinges on a specific pair of proteins found in the blood. By pinpointing these biological signatures—a process often categorized in the field of proteomics—the test can distinguish between a healthy individual and a patient with a malignant growth. Dr. Simon Newman, chief scientific officer at the Brain Tumour Charity, characterizes the research as a pivotal milestone:
“The research marked a significant step towards a simple blood test that could help clinicians detect glioblastoma.”
The Expert Take While a neurosurgical biopsy remains the definitive “gold standard,” it is a resource-heavy intervention requiring a surgical theatre, a neurosurgeon, and an array of support staff, costing thousands of pounds per patient. In contrast, a phlebotomy appointment takes minutes. Replacing even a fraction of initial biopsies with a 90% accurate blood test would not only mitigate the physical risks of brain surgery but also drastically reduce the financial and logistical strain on oncology departments.
3. From Surgery to “Covid-Style” Simplicity
The project’s lead, Prof. Petra Hamerlik, isn’t just looking for a lab breakthrough; she is aiming to democratize the diagnostic process. Her vision is to condense this complex proteomics research into a device with “Covid-style” simplicity—a tool that could sit on the desk of every GP in the country.
The proposed workflow would revolutionize the “triage” process. Currently, many patients face a tragic “diagnostic lag.” In one instance cited in recent reports, a patient noted that doctors “weren’t worried” about their symptoms until it was too late. This new test provides GPs with an objective metric:
- A patient presents with recurring headaches (the “three visits” threshold).
- The GP administers the blood test in-clinic.
- The device generates a “risk score” based on the protein pair detection.
- Only those with a high-risk score are fast-tracked for an urgent MRI.
The Expert Take This “risk score” methodology acts as a vital gatekeeper for the NHS’s overburdened imaging infrastructure. By filtering out low-risk cases at the primary care level, the test ensures that limited MRI slots are reserved for those in genuine peril, effectively shortening the wait time for patients who need immediate intervention.
4. Monitoring the “Devastating” Enemy in Real Time
Glioblastoma is what Prof. Hamerlik describes as “one of the most devastating cancers we face.” Its primary weapon is its speed and its tendency to recur. Because the Manchester study validated the blood test using patients already undergoing surgery, radiotherapy, and chemotherapy, the test is uniquely suited for longitudinal monitoring.
Prof. Hamerlik explains the necessity of catching these signals early during the initial diagnostic phase:
“The idea with our work is that, if you come more than once with a headache, such as three times, and if the doctor suspects a brain tumour, they would run this test. The risk score would indicate that you might have a malignant growth in your brain, and you should go for MRI.”
This same risk-score mechanism allows the test to monitor the tumor’s response to treatment. If the protein levels shift during chemotherapy, the test provides a real-time indicator of efficacy.
The Expert Take In modern oncology, we often rely on “snapshots”—an MRI scan taken every few months. However, glioblastoma doesn’t wait for scheduled appointments. Transitioning to a “real-time” data stream through regular blood draws allows clinicians to observe the tumor’s behavior continuously. This is the difference between seeing a few still frames and watching the whole movie; it allows for more agile treatment adjustments that could potentially extend lives.
5. The Personal Mission and the Global Trial
The drive behind this breakthrough is more than academic; it is fueled by a personal vendetta against the disease. Prof. Hamerlik’s father was himself a glioblastoma patient, a fact that infuses the research with a rare level of urgency. This disease is notoriously indiscriminate, a reality highlighted by the high-profile death of The Wanted singer Tom Parker, who passed away in 2022 at just 33 years old.
The project has now graduated to a massive validation phase to ensure its global applicability. Clinical trials are currently active across ten international locations:
- Six sites across the United Kingdom.
- Four sites abroad, including the initial collaborative teams in Denmark.
The Expert Take Scientific breakthroughs are often viewed as sterile, data-driven events, but the most rigorous advancements frequently stem from personal stakes. When the lead researcher has witnessed the “devastating” reality of the disease in her own home, the commitment to making the technology both accessible and infallible becomes the primary driver.
6. The Roadmap to the NHS: A Decade of Development
Despite the 90% accuracy already achieved, the test faces a long road before it becomes a staple of the NHS. Researchers estimate a timeline of roughly a decade for full integration. The roadmap includes:
- Mass Validation: Confirming the protein signatures across diverse global populations.
- Regulatory Hurdles: Navigating the stringent safety and efficacy standards of healthcare regulatory bodies.
- Expansion: Training the test to identify a broader spectrum of brain tumor types beyond glioblastoma.
The Expert Take A ten-year horizon may seem slow to a patient in need, but it reflects the reality of the “valley of death” in med-tech—the gap between a successful lab trial and a safe, scalable clinical product. This decade will be spent gathering longitudinal data to ensure the “risk score” is a bulletproof metric, as a false negative in brain cancer is a death sentence, while a false positive leads to unnecessary, high-risk neurosurgery.
7. Conclusion: A New Era of Early Detection
The Manchester study signals the beginning of the end for the “wait and see” era of brain cancer. By trading the complexity of a surgical biopsy for the simplicity of a blood draw, the medical community is moving toward a proactive model where the most aggressive cancers are caught before they become terminal.
The shift from invasive diagnostics to “Covid-style” ease at the GP’s office could revolutionize the standard of care for thousands of families.